Paraproctitis occurs regardless of age, but more often in men, because they smoke a lot and drink often. However, recently more and more women and girls are becoming ill: the desire to wear tight-fitting trousers, especially jeans made of thick, coarse fabric, as well as underwear, which, cutting into the crotch, injures the skin, is affected. Such abrasions become susceptible to infection, including viral infections. When hypothermia occurs - this is when girls flaunt in short skirts and thin tights in cold weather - herpes appears, which also provokes paraproctitis. Elderly people usually suffer from chronic paraproctitis if they suffered an acute form of the disease in their youth.
Causes of inflammation of the rectum
Diseases may be of an allergic nature. But more often they are caused by opportunistic bacteria, which are constantly present inside the organ and lead to inflammation only in the presence of provoking factors. Less commonly, viruses that enter the intestinal tissue with the blood. The inflammatory process can be a consequence of sexually transmitted infections.
Factors provoking pathologies:
- frequent constipation/diarrhea;
- poor nutrition;
- weakened immune system;
- undergoing radiation therapy;
- taking antibacterial drugs.
The diseases often occur in patients suffering from food allergies, Crohn's disease, and ulcerative colitis. Hemorrhoids and anal fissures contribute to their occurrence. Pathologies can develop as a complication of sepsis, influenza or sore throat.
Paraproctitis - what antibiotics to take?
The choice of antibiotics is made by a doctor based on the suspected causative agent of the infectious process (empirically) or the data obtained from a bacteriological study with determination of the sensitivity of the isolated pathogen to the main groups of antibacterial agents. Today the following groups are most often used:
- Semi-synthetic penicillins.
- Cephalosporins.
- Macrolides.
They can also be used in various dosage forms. Depending on the severity of the inflammation, these can be suppositories with antibiotics for paraproctitis , tablets or capsules, as well as a parenteral form of the drug (injections).
If, based on clinical, laboratory and instrumental examination data, paraproctitis has been diagnosed, which antibiotics to use, their dosage and the duration of the course of their use are determined by the attending physician. Independent and uncontrolled use of antibacterial agents is fraught with the subsequent development of complications.
Symptoms of acute proctitis and paraproctitis
Pathologies develop as an acute inflammatory process. Its clinical picture is identical in both cases. This is due to the close proximity of the mucous membrane and fiber of the rectum.
Local symptoms
- constipation/diarrhea;
- fecal incontinence;
- discharge of mucus, pus, and sometimes blood from the anus;
- a feeling of empty bowels and a constant desire to go to the toilet;
- itching and pain around the anus, aggravated by defecation;
- pain on the left side of the abdomen, in the perineum (sometimes radiating to the genitals or lower back).
General symptoms
- weakness;
- headache;
- skin rashes;
- loss of appetite;
- weight loss;
- aches and pain in muscles;
- increase in body temperature to +38…+39 oC, chills.
On methods of diagnosis and conservative treatment of anorectal diseases
Despite the fact that anorectal pathology has long attracted the attention of researchers, until now it has received insufficient attention. Basically, surgical methods of treatment are described, to the development of which A. V. Vishnevsky, A. N. Ryzhikh, M. Kh. Levitan, Zh. M. Yukhvidova, V. D. Fedorov, G. I. Vorobyov made a significant contribution.
The purpose of our article is to present traditional and modern methods of diagnosis and conservative treatment of anorectal diseases.
Diagnostics
Since ancient times, methods such as examination of the anus and digital examination of the rectum have been used to diagnose anorectal pathology. Later, rectoscopy began to be used. These techniques have not lost their importance to this day.
Examination of the anus is the first stage of examination if anorectal pathology is suspected. The method makes it possible to identify external hemorrhoids and hemorrhoidal fimbriae, external openings of fistulas, and anal fissures. With idiopathic anal itching and proctitis, weeping and excoriation of the skin around the anus can be observed; with paraproctitis localized in the subcutaneous tissue, hyperemia and swelling of the perianal skin can be observed. When examining the anus, you can see an anal polyp, prolapse of internal hemorrhoids, the mucous membrane of the anal canal, or all layers of the rectum.
Digital examination has always been and remains a mandatory part of a proctological examination. The importance of this method was repeatedly emphasized in his speeches by Academician V. Kh. Vasilenko. He was supported in this by Professor V.M. Mysh, who wrote: “The scope of diagnostic capabilities of sigmoidoscopy is limited to diseases of the intestine itself, while digital examination through the rectum is an extremely valuable and widely available method for studying both the intestine itself and a number of adjacent organs.” In the diagnosis of rectal cancer, digital examination is of particular importance and makes it possible in 88% of cases to detect a tumor, determine the degree of its displacement, the distance from the anus (which is important for choosing the method of surgical intervention), and the narrowing of the intestinal lumen. In addition to examining the rectum, this method determines the condition of neighboring organs (prostate gland in men, cervix and posterior surface of the uterine body in women).
Anoscopy - examination of the anal canal and lower ampullary part of the rectum using rectal mirrors is also included in the group of methods for mandatory examination of patients with anorectal pathology. During this examination, hemorrhoids can be identified, true polyps can be differentiated from hypertrophied anal papillae, which are hyperplasia of the mucous membrane in the area of Morganian crypts, resulting from chronic inflammation in anal fissures, hemorrhoids or proctitis.
Sigmoidoscopy has long been considered the main method for visual examination of the rectum and distal sigmoid. In subsequent years, the rigid sigmoidoscope was replaced by flexible fiberglass sigmoidoscopes, which make it possible to examine the entire left part of the colon, the most common location of tumors.
Total colonoscopy is performed in cases where sigmoidoscopy reveals polyps or malignant tumors in the rectum or sigmoid colon, when an inflammatory process is detected that is not limited to the distal colon, or there is a pathological discharge (mucus, blood, pus) in the intestinal lumen coming from proximal sections.
Ultrasonography. Currently, to recognize complicated rectal pathology, ultrasound devices equipped with rectal sensors have begun to be used, which makes it possible to obtain information about the transmural spread of the inflammatory process in the distal colon, the presence of infiltrates, fistulas and abscesses.
Thus, today colonoscopy and ultrasound using rectal sensors have joined the arsenal of the above-described and well-known diagnostic methods.
Characteristics of anorectal diseases
Hemorrhoids are one of the most common diseases; it affects 10% of the population. Among proctological diseases, hemorrhoids account for 40%.
The views of researchers on the etiology of hemorrhoids are contradictory. Congenital insufficiency of the venous system, venous stagnation, constipation, and disruption of the rectal sphincter mechanism were cited as causative factors. At the same time, none of the hypotheses based on the pathology of the venous system for a long time could explain the origin of the main symptom of hemorrhoids - the release of scarlet blood. In 1975, W. Thomson experimentally proved the existence of arterial and venous components of the hemorrhoid [9]. He formulated the cause of hemorrhoids as a primary weakness of the epithelium of the anal canal, leading to slipping and displacement of the anal “cushions,” which can occur with chronic constipation or prolonged straining during bowel movements [10].
Summarizing the research conducted to study the pathogenesis of hemorrhoids, G. I. Vorobyov and co-authors came to the conclusion that “the leading factors in the pathogenesis of hemorrhoids are dysfunction of the vessels that provide blood flow through the cochlear arteries and outflow through the efferent veins” [1].
There are several classifications of hemorrhoids [1, 4–6].
According to one of the latest classifications, hemorrhoids are divided according to their clinical course into acute and chronic, and according to their form into internal, external and combined. The chronic course of hemorrhoids, in turn, is divided into four stages: Stage I - discharge of blood from the anus without prolapse of hemorrhoids; II - loss of nodes with spontaneous reduction into the anal canal (with or without bleeding); III - periodic prolapse of nodes with the need for their manual reduction into the anal canal, stage IV - constant prolapse of hemorrhoids along with the rectal mucosa [1].
Harbingers of hemorrhoids over a long period of time (from several months to several years) may be discomfort in the anus or anal itching. The first and main symptom of the disease is anorectal bleeding of varying severity - from scanty bloody traces on toilet paper or stool to massive bleeding, leading to anemia in 1% of cases. The blood is usually bright red, but may be dark if it accumulates in the rectal ampulla. At the very beginning of the act of defecation, blood accumulated in the rectum can be released in the form of clots. More often, patients note the release of blood in the form of drops or a splashing stream. Occasionally, bleeding is observed outside the act of defecation. Hemorrhoids are also characterized by pain in the anus, which occurs during bowel movements, walking, and poor diet (eating spicy foods, alcoholic drinks). The cause of pain may be changes in the perianal area with external hemorrhoids or associated complications (anal fissure, thrombosis of the external hemorrhoidal plexuses). Anal itching occurs quite often with hemorrhoids and is a consequence of excessive mucus secretion, contamination of the anal area with blood and fecal particles. This constantly causes a feeling of wetness around the anus and soiling of the underwear. As a result, scratching appears and excoriation of the skin of the perianal area occurs (Fig. 1).
At the initial stages of the disease, conservative therapy is carried out. Attention is paid to nutrition. Alcoholic drinks and irritating foods contribute to increased hemorrhoidal bleeding, so alcohol, seasonings, spicy and salty foods are excluded from food. After defecation and toileting the anus, soft suppositories are inserted into the anus. For a long period of time, in proctological practice, suppositories of the following composition have been deservedly used for these purposes: Extr. Belladonnae 0.015; Novocaini 012; Xeroformi 0.1; But. Cacao 1.7. In case of bleeding, add S. Adrenalini 1:1000 gtt to the above composition. IY. Other candles listed below are currently in use.
In acute hemorrhoids and hemorrhoids complicated by thrombosis of the hemorrhoid, conservative therapy is first carried out, aimed at eliminating the inflammatory process and regulating stool. On the first day, cold is prescribed to the perineal area, in subsequent days - warm sitz baths with a weak solution of manganese after stool and rectal suppositories of the above composition, as well as suppositories or ointment Ultraproct, Posterizan, Proctoglivenol, Relief, Relief Ultra or Advance. These drugs are prescribed rectally in the form of cream or suppositories 2 times a day after stool and at night. In addition to local therapy, drugs that have a venotonic and angioprotective effect are prescribed orally. One of these drugs, Detralex, belongs to the group of flavonoids and is a combination of diosmin and hesperidin. The drug helps reduce the distensibility of veins, increase their tone, reduce stagnation and improve microcirculation. Detralex is prescribed 6 tablets per day for the first 4 days, then 4 tablets per day for the next 3 days.
The second drug in the same direction, Phlebodia 600, is non-micronized diosmin. The drug is prescribed according to the following regimen: 2–3 tablets per day with meals for 7 days, then, if necessary, treatment can be continued at 1 tablet 1 time per day for 1–2 months. For the same purpose, Diovenor 600, an analogue of Phlebodia 600, is used.
In case of prolapse of nodes, frequent exacerbations that do not respond to conservative therapy, and heavy recurrent bleeding, surgical treatment is indicated (Fig. 2). According to the Mayo Clinic, the most satisfactory results were obtained by alloying the nodes with a latex washer and hemorrhoidectomy.
Anal fissure ranks second among anorectal diseases and is a linear or triangular mucosal defect 1.0–1.5 cm long, located near the transitional fold, above the Hilton line. The origin of the fissure is associated with many reasons, but the most important factor is trauma to the mucous membrane of the anal canal by feces, foreign bodies, and damage to it during childbirth. A predisposing factor may be hemorrhoids.
A sharp crack has a slit-like shape, smooth, even edges. Its bottom is made up of sphincter muscle tissue. With a long course of the pathological process, connective tissue grows along the edges of the crack. Its bottom is covered with granulations. In the area of the outer edge of the crack, excess tissue forms the anal (sentinel) tubercle. Thus, an acute fissure turns into a chronic one, which is an ulcer with scarred edges and a scarred bottom (Fig. 3).
Sometimes an acute crack heals on its own, but more often it becomes chronic. As a rule, there is only one crack and most often it is located on the back wall of the anal canal. Occasionally you can observe two cracks that are located one above the other. It is necessary to distinguish true cracks from various superficial damage to the perianal skin. The most common localization of cracks (at 12 o'clock on the posterior wall and at 6 o'clock on the anterior wall) is explained by the structural features of the anal sphincter. It is at 6 and 12 o'clock that the blood supply conditions are worse, and there is a great danger of injury to the mucous membrane during the passage of feces during defecation due to pressure on the posterior and anterior commissures. Trauma to the mucous membrane leads to acute pain due to irritation of numerous nerve endings and sphincter spasm. A vicious circle is formed - an anal fissure leads to a sharp pain syndrome, pain syndrome leads to sphincter spasm, sphincter spasm prevents the healing of the fissure.
A triad of symptoms characteristic of the clinical picture of an anal fissure is pain during and especially after defecation, sphincter spasm and scanty blood discharge. There are differences in the clinical picture of acute and chronic fissures. With an acute fissure, the clinical manifestations are pronounced. The main symptom is acute pain after defecation, lasting several hours. Sphincter spasm increases pain. The pain becomes throbbing. Blood is detected as droplets on the surface of the stool. Due to sphincter spasm and a feeling of fear before defecation, stool is delayed. Taking laxatives increases pain.
With a chronic fissure, the pain is less acute, its duration after stool is 5–10 minutes. Sphincter spasm is mild. When a chronic anal fissure is complicated by marginal fistulas, purulent discharge, itching, and irritation of the perianal skin appear. A chronic crack is characterized by a cyclic course. It can heal, but at the slightest strain or physical stress it opens again.
Detecting an anal fissure is not difficult. Anamnesis and characteristic complaints suggest its existence. The diagnosis becomes clear already upon examination of the anus. When spreading the buttocks, a crack or guard tubercle is discovered at its outer edge. If the crack is not visible upon examination, then a digital examination of the rectum should be carefully performed. In this case, a compaction will be detected on the affected wall of the anal canal. Sigmoidoscopy in the acute period should not be performed due to severe pain and sphincter spasm. In cases where it is necessary to differentiate an anal fissure from other diseases, the study can be carried out after a preliminary injection of 4-5 ml of 1% novocaine solution under the fissure. Differential diagnosis is carried out with coccydynia and the anal form of Crohn's disease.
Conservative treatment of an acute fissure involves, first of all, eliminating pain, sphincter spasm and normalizing stool. To normalize stool, Mucofalk can be used, containing hydrophilic fibers from the outer shell of plantain seeds that retain water. As a result, the volume of stool increases and its consistency becomes softer. The drug is well tolerated by patients and does not increase pain in the abdomen and anus, which is characteristic of many laxatives. A diet limiting spicy and irritating foods is recommended. Once a day, cleansing enemas of boiled water at room temperature are given. In this case, the enema tip is thickly lubricated with petroleum jelly and inserted along the edge of the anus opposite to the one along which the crack is located.
After cleansing the intestines, take warm (38 °C) sitz baths with a weak solution of manganese for 10 minutes. Self-stool should be avoided during this period due to the risk of increased pain and spasm. Laxatives, as mentioned above, are contraindicated, as they can increase pain. Then soft-based suppositories (Relief, Relief Advance, Ultraproct, Posterisan) are inserted into the rectum. Treatment with suppositories is carried out until the crack is completely epithelized. Cleansing enemas, as a rule, are given during the first 10 days from the onset of the disease, after which the stool is independent.
When conservative therapy is ineffective, surgical treatment is resorted to, which consists of excision of the fissure and sphincterotomy. In recent years, closed lateral subcutaneous sphincterotomy has been proposed for the treatment of uncomplicated anal fissure [3, 7], after which pain disappears faster and there are fewer relapses.
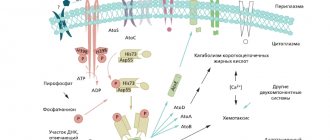
Paraproctitis is inflammation of the tissue surrounding the rectum. Among proctological diseases, paraproctitis accounts for 15%. There are three fascial-cellular spaces: subcutaneous, ischiorectal and pelvic rectal. Accordingly, paraproctitis is divided into subcutaneous, submucosal, ischiorectal and pelvic-rectal. The disease is caused by various microorganisms that penetrate into the cellular spaces from the rectum through the anal glands, damaged mucous membrane, as well as hematogenously or lymphogenously from neighboring organs affected by the inflammatory process.
Of particular importance in the pathogenesis of paraproctitis is direct damage to the mucous membrane of the rectum in the area of the posterior wall of the anal canal, where wide and deep crypts are located, which are the entrance gates of infection. From 6 to 8 ducts of the anal glands open into each crypt. Through them, the infection spreads to the perirectal tissue spaces. According to the clinical course, paraproctitis is divided into acute and chronic. Acute paraproctitis is a purulent inflammation (abscess) of the peri-rectal tissue. Chronic paraproctitis is a consequence of acute inflammation. This is a pararectal fistula, formed after spontaneous opening of an abscess or opening it surgically. The internal opening of a pararectal fistula is a defect in the rectum. The external opening is located on the skin of the perineum (Fig. 4).
In some cases, several fistula tracts and several external openings of the fistula can be observed. The disease begins with a prodromal period (up to three days), during which weakness, headache, and general malaise may be observed. Then chills, fever, and pain in the perineal area appear. When the cellular spaces of the small pelvis are affected by the type of phlegmon, the clinical picture is dominated by general symptoms caused by intoxication. As the process is limited and an abscess is formed, the intensity of the pain increases. It takes on a pulsating character.
The greatest importance in the diagnosis of acute paraproctitis is examination of the anal area and digital examination of the rectum. Upon examination, attention is drawn to hyperemia of the skin in the perineal area on the affected side. Palpation of the perineum is painful, fluctuation can be detected. Digital examination is also painful in many cases and should be performed with caution. This technique should not be neglected, since the information obtained through digital examination can be very valuable for recognizing almost all forms of acute paraproctitis.
Treatment of acute paraproctitis, carried out immediately after diagnosis, is surgical. Basic principles of treatment: 1) opening the abscess; 2) elimination of the internal opening through which the abscess cavity communicates with the rectum.
Chronic paraproctitis, as mentioned above, is a consequence of acute paraproctitis. If, when opening the abscess, the internal opening in the rectum is not eliminated, a rectal fistula may subsequently form. A rectal fistula is characterized by the presence of one or more external openings in the skin of the perineum. Pus, feces and gases may be released from the external openings of the fistula tract. The general condition of patients with chronic paraproctitis suffers little. Pain appears only when the process worsens. Depending on the location of the fistula tract in relation to the sphincter muscle, four types of fistulas are distinguished: subcutaneous-submucosal (intrasphincteric), transsphincteric, complex (extrasphincteric) and incomplete. The latter have only an internal opening in the rectum and do not have an external opening in the perineum. The source of their formation is often an anal fissure.
In the diagnosis of chronic paraproctitis, the same methods are used as for acute paraproctitis. Digital examination of the rectum makes it possible to judge the tone of the sphincter and, in some cases, detect the internal opening of the fistula. Be sure to perform probing with a button probe, which is inserted into the fistula tract through its external opening (Fig. 5).
Using a probe, the direction of the fistula tract and its relationship to the sphincter muscle are determined. Fistulography is a mandatory x-ray examination of rectal fistulas. A test with a dye is used to determine the patency of the fistula tract, the location of the internal opening and purulent cavities in the tissue. Sigmoidoscopy is performed to detect concomitant diseases and high-lying internal fistula openings.
Treatment of all types of fistulas is surgical. The operation can be considered radical only if the opening in the rectum is eliminated by excision of the fistula tract.
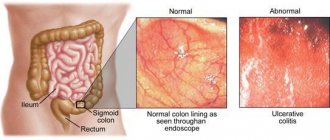
Proctitis is an acute or chronic inflammation of the mucous membrane of the rectum or rectum and sigmoid colon (proctosigmoiditis). Acute proctitis most often has a specific cause. It can be caused by trauma, foreign bodies, mechanical irritation during prostate massage, chemical and thermal burns, and frequent cleansing enemas. The disease may be a consequence of radiation therapy for malignant tumors of the pelvic organs (radiation proctitis) or infections (gonorrheal, dysenteric proctitis). The development of proctitis can be facilitated by diseases of neighboring organs (hemorrhoids, anal fissure, paraproctitis, Douglas pouch abscess, prostatitis, cystitis, vulvovaginitis, etc.). Long-term proctitis and proctosigmoiditis that cannot be treated conservatively should, first of all, be differentiated from distal forms of ulcerative colitis [2].
Acute proctitis has a sudden onset, may be accompanied by fever, chills, is characterized by inflammatory changes in the rectal mucosa, clinically manifested by a frequent urge to defecate, tenesmus due to constipation, a burning sensation and a sensation of a “foreign body” in the anus. Examination of the rectum with a finger and sigmoidoscopy are poorly tolerated by patients due to severe pain. The rectal mucosa is sharply hyperemic, swollen, and bulges into the lumen. The vascular pattern is blurred or absent.
Chronic proctitis, as a rule, is a consequence of an untreated acute disease or has a specific nature. Clinically, the disease is manifested by a periodic feeling of discomfort in the rectum, a feeling of incomplete emptying, periodic exacerbations, accompanied by frequent stools mixed with mucus (sometimes blood), and tenesmus. Endoscopy reveals hyperemia of varying severity and swelling. The diagnosis requires the exclusion of infectious, parasitic and other diseases.
Treatment of acute proctitis is conservative. First of all, you need a diet that excludes all irritating foods and alcohol. After complete cleansing of the intestines, 100.0 ml of warm chamomile infusion (temperature 37–38 °C) is injected into the intestine for therapeutic purposes. An oil enema is given at night (50–75 ml of warm -37–38 °C vegetable oil). Starting from the second week of the disease, morning medicinal chamomile enemas are replaced with enemas of 0.3–0.5% collargol solution. The concentration of the solution is determined by the intensity of the inflammatory process in the intestine. Oil evening microenemas are continued for 14 days. The general course of treatment is 2–3 weeks. After a 10-day break, the course of treatment should be repeated to avoid relapse.
When treating chronic proctitis, the same remedies are recommended as for acute proctitis, but the course of conservative therapy is longer. The best effect is achieved by therapeutic microenemas from a collargol solution.
Anal itching is a pathological condition characterized by persistent scratching in the anus. It is necessary to clearly distinguish idiopathic (primary) anal itching and secondary, often accompanying diseases such as hemorrhoids, anal fissure, helminthic infestations, proctitis and proctosigmoiditis. While the causes of secondary anal itching are known, the etiology of primary (idiopathic) itching is not fully understood.
Often itching in the anus is the patient's main and only complaint. According to the clinical course, acute and chronic anal itching are distinguished. The acute form is characterized by a sudden onset, a constant course, significant intensity and local changes in the skin such as wet eczema with maceration, traces of scratching, hypertrophy of the perianal folds. With chronic itching, on the contrary, the onset is slow, the skin is often dry, thinned, sometimes depigmented, there are no traces of scratching at all or they look like thin linear abrasions.
Diagnosis of the causes of anal itching always begins with determining the type of itching (idiopathic or secondary). First of all, it should be determined whether itching is a symptom of the above diseases (secondary), whether its occurrence is associated with the intake of spicy or salty foods, alcohol, contact with harmful chemicals and radioactive substances, the presence of allergies, etc. From laboratory tests, a blood test is performed for sugar content and three-time stool analysis for worm eggs. In men, urethritis and prostatitis should be excluded; in women, vaginal itching should be excluded.
Treatment of secondary anal itching consists of treating the underlying disease. For idiopathic anal itching, treatment is purely symptomatic.
A diet excluding spicy foods and alcohol is recommended. Thorough toileting of the anus after each act of defecation. After this, ointments (Posterizan, Proctosedyl, Aurobin, Emalan) are injected into the rectum. At night, warm sitz baths with a solution of manganese and microenemas from a 0.33% solution of collargol -50.0 ml are prescribed. The course of treatment is 10 days. For the next 10 days, microenemas (50 ml) of warm vegetable oil are prescribed.
Methods of conservative treatment of anal pathology are being improved as new drugs are developed. From this point of view, one should pay attention to medical collagens, which have become used in the complex therapy of many diseases.
The problem of studying collagen as a new plastic material has long attracted the attention of researchers. At the origins of the development of this direction was Academician of the USSR Academy of Medical Sciences V.V. Kovanov. As a result, a new class of medicinal dosage forms was created - plastic materials with targeted action based on the natural biopolymer of collagen. Currently, medical collagen Emalan, which is a collagen hydrogel for problematic skin and wounds, has become widely used in practical healthcare. It successfully passed clinical trials at the Burn Center of the Research Institute of Emergency Medicine named after. N.V. Sklifosovsky (1995), at the Military Hospital named after. N. N. Burdenko (1999) and in the 72nd Central Clinic of the Ministry of Emergency Situations (2009). It has been established that Emalan reduces signs of inflammation. Under its influence, hyperemia, swelling, pain disappear, itching and peeling of the skin decreases or disappears. All of the above served as the basis for the use of this drug in the treatment of anal pathology.
Of the 112 we observed during 2009–2010. 54 patients had acute and chronic anal fissures, 50 had grade I and II hemorrhoids, and 8 had idiopathic anal itching.
When treating anal fissures, Emalan was included in complex therapy. It was introduced into the rectum on a suppository and in the form of applications to the anus for 3 weeks. In 70.3% of cases, positive dynamics were noted already on days 4–5 from the start of treatment. By the end of the third week, 31 patients had complete healing of the fissures. In patients suffering from hemorrhoids complicated by anal fissure and itching (50), by the end of the third week, against the background of complex therapy, a decrease in symptoms of the disease, such as itching, pain, burning, was observed. The drug turned out to be especially effective in the treatment of patients with idiopathic (8) and secondary (6) anal itching. While taking the drug (gel applications to the anal area 3-4 times a day), a decrease in scratching, maceration and skin hyperemia was noted. By the end of the two-week course, these changes completely disappeared.
Our first experience has shown that the domestic collagen gel Emalan can be successfully used in the complex treatment of acute and chronic anal fissures, proctitis, anal itching and complicated forms of hemorrhoids. The drug makes it possible to increase the effectiveness of therapy and helps reduce treatment time.
Literature
- Vorobyov G.I., Shelygin Yu.A., Blagodarny L.A. Hemorrhoids. M. // "Mitra-Press". 2002. 192 p.
- Grigorieva G. A. Anorectal pathology. Guide to gastroenterology // M.: Medicine. 1996. T3. 606–635 pp.
- Henry M. M., Swash M. Coloproctology and the pelvic floor // M.: Medicine. 1988 (translation from English). 451 p.
- Rivkin V.L., Kapuller L.L. Hemorrhoids // M.: Medicine. 1984. 751 p.
- Rivkin V.L., Dultsev Yu.V., Kapuller L.L. Hemorrhoids and other diseases of the anal canal // M.: Medicine. 1994. 239 p.
- Rivkin V. L., Kapuller L. L. Hemorrhoids. Constipation // M.: Medpraktika. 2000. 158 p.
- Fedorov V.D., Dultsev Yu.V. Proctology. M.: Medicine. 1984. 380 p.
- Haas PF, Fox TA, Haas GP The pathogenesis of haemorrhoids. Disease of the colon and rectum. 1984. T. 27. p. 442–450.
- Thomson WHF The nature of haemorrhoids // British J. of Surgery. 1975. T. 62. p. 542–552.
- Thomson WHF The nature and cause of haemorrhoids. Proceedings of the Royal Society of Medicine. 1975. T. 68. p. 574–575.
G. A. Grigorieva , Doctor of Medical Sciences, Professor S. V. Golysheva First Moscow State Medical University named after. I. M. Sechenova , Moscow
Contact information for authors for correspondence
Possible consequences
Lack of treatment for acute proctitis leads to the disease becoming chronic. Against this background, anal fissures often appear, and the inflammation spreads to the fatty tissue and causes paraproctitis. All this provokes the formation of malignant tumors in the tissues of the rectum.
Paraproctitis is accompanied by the formation of a large amount of pus. This usually leads to the formation of a fistula, which finds its outlet near the anus. Sometimes the abscess breaks through. Then the purulent masses spread in the abdominal cavity, causing peritonitis and involving other organs in the inflammatory process. This can lead to blood poisoning (sepsis).
Types of operations to remove acute purulent paraproctitis
In acute ischiorectal paraproctitis, when the primary fistula is located trans- or intrasphincterically, the superficial structures of the sphincter are involved
In this case, an autopsy of acute paraproctitis is performed, and the primary purulent tract is excised into the lumen of the rectum. Stepping back at least 3 cm from the edge of the anus, an incision about 6 cm long is made. The pus is evacuated, the purulent cavity is sanitized. A probe is inserted into the intestinal lumen through the primary purulent tract, the mucous membrane is dissected along it and excised in the shape of a triangle with its apex in the anal canal. After treating the abscess cavity, a gas outlet tube is inserted into the rectum and tamponade is performed.
When the primary fistula is located transsphincterically, a significant part of the sphincter is involved
Surgical intervention includes two stages:
- Opening and draining the abscess;
- On days 5-6 - closing the internal opening in the wall of the rectum by moving the mucous membrane. The flap of fabric is fixed with sutures.
At the end of the operation, a gas tube is inserted into the rectum and tamponade is performed.
In case of exacerbation of recurrent paraproctitis when the purulent tract is located extra- or transsphincterically
Paraproctitis opens into the lumen of the rectum. On days 5-6, the internal opening is closed with a mucomuscular flap. If the scarring process is widespread along the anal canal and the mucous membrane is motionless, a ligature is made.
How are pathologies treated?
Despite the similarity of clinical manifestations, proctitis and paraproctitis are treated differently. Treatment of inflammation of the mucous membrane is conservative. If the inflammatory process affects the intestinal tissue, surgical intervention is required.
Conservative therapy
The goal of therapy is to stop the inflammatory process. For this, the patient is prescribed antibiotics (Ceftriaxone, Medaxone) and antimicrobial agents (Biseptol, Bactrim). To rinse the rectum, enemas are done with Collargol, furatsilin solution or warm chamomile infusion. When the symptoms of inflammation decrease, sitz baths in a weak solution of potassium permanganate and oil microenemas are additionally prescribed.
Treatment of proctitis includes diet. The patient is recommended to exclude fatty, spicy, spicy foods, alcohol, and high-fiber foods from the diet. After recovery, he is advised to consume more dietary fiber along with fruits, vegetables and grains to prevent constipation.
Surgical intervention
Paraproctitis requires emergency surgery. It is carried out immediately after diagnosis using general anesthesia. During this procedure, the abscess is opened, its contents are removed, and the cavity is washed with an antiseptic solution.
To prevent relapse and complications, the operated patient is prescribed a course of antibiotics. Wounds are treated daily with antiseptics or antimicrobial agents. In the postoperative period, it is necessary to follow a diet. It prohibits the consumption of foods that cause gas formation and contain large amounts of fiber, spicy and fatty foods.
If symptoms of proctitis and paraproctitis appear, contact your doctor. An experienced proctologist will see you, make an accurate diagnosis and prescribe effective treatment. If necessary, you will receive emergency care immediately after diagnosis.
Treatment of paraproctitis
The most common treatment for paraproctitis is ichthyol ointment and suppositories. They reduce tissue tension, relieve pain, help patients adapt to their condition; some even refuse surgery.
Initially, we try to puncture the abscess - release the pus, and then relieve the inflammation. The fistula is washed with a 1% dioxidine solution, and at the same time the patient is prescribed an anti-inflammatory drug - sumamed (0.5 g once a day for 3 days in a row). Fistula lavage is performed on an outpatient basis. Typically, after three procedures, the fistula closes in most patients. This is followed by taking medications that support the immune system.
But it happens that the fistula remains, however, without obvious purulent discharge. Treatment continues at home. Sitz baths are recommended: 1 tbsp. spoon of sea salt and baking soda per 5 liters of warm water. The duration of the bath is 10 minutes. Such procedures help the discharge of pus and clean the fistula well. After the bath, suppositories or ultraproct ointment are introduced. You can make microenemas from mumiyo: dissolve 1 tablet in 0.5 cups of hot water, administer warm.
I’ll tell you about one amazing case of recovery, which was helped by... a honey fair organized by Moscow Mayor Yuri Luzhkov in Manege. The patient came to us for procedures for a whole month, and to no avail: there was no pus, but the fistula remained. Here we just received information from this fair about the healing properties of sweet clover honey. They began to give the patient microenemas - 1 tbsp. a spoonful of honey in 0.5 cups of water. And everything healed in two days. Just a miracle!
I must note that such enemas - both with honey and with propolis oil solution - are very effective. The intestine even begins to squelch with pleasure: she likes warm, sweet things so much! Microclysters (1 tablespoon per 0.4-0.5 glasses of water) are done at night, and in the morning after stool, UltraProct suppositories are administered.
Sometimes you have to wait a long time for the abscess to break through. Then we recommend using the same ichthyol ointment, cakes made from baked onions, potatoes, and carrots. They help the fistula break through and pus drain. The patient chooses which flatbread is better, as they say, “at random” - whichever works more effectively, he “cooks” from it.
When all methods of conservative treatment have been exhausted, but the disease does not go away, we refer the patient for surgery, of course, if there are no contraindications. It is called Gabriel's operation, named after the surgeon who first performed it: a speculum is inserted into the anus, the mucous gland and fistula are excised. The treatment is very radical. True, scars appear after it, and even with hard stool, the lack of mucus causes certain inconvenience. Therefore, we try to injure this area as little as possible and, if possible, operate in extreme cases. It is better to use folk remedies and be observed at the clinic for a year. If the fistula heals, you can forget about the operation for decades.